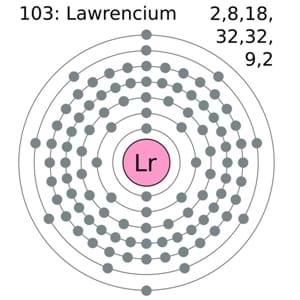
Wondering about the Lawrencium valence electrons? Well, we are here to disclose all the significant aspects of these chemical elements along with their valency and valence electrons in Periodic Table. All our chemistry scholars can find the significant properties and other characteristics of this chemical element for their general and academic knowledge. Lawrencium is the chemical element that comes with its atomic number 103 and the symbol of Lr. The element is basically the member of the Actinide series of the element and has high radioactive chemical properties. It was named back in the honor of Ernest Lawrence during the 1950 and remains mostly in its research phase.
How Many Valence Electrons Does Lawrencium Have?
When we talk about the valence electrons of any element then we simply refer to the electrons of such elements. These electrons actually hold their space in the outer shell of the element for the atom. The valence electrons have their contribution in the combining process of the element with the other elements.
So, in the case of the Lawrencium, we have the 3 (Three) valence electrons in the outer shell of the element. You can refer to the periodic table of the element in order to find more information on the same aspect.
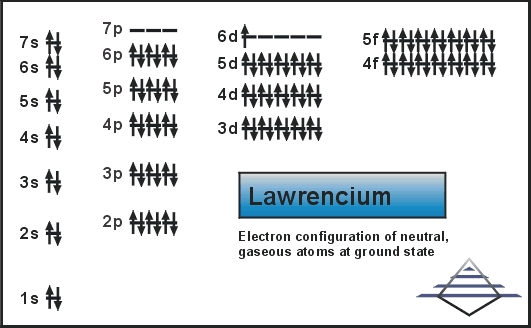
Lawrencium Valence Electrons Dot Diagram
Well, if you are seeking the diagrammatical representation for the valence electrons of the Lawrencium then you can simply refer to its dot diagram. This particular dot diagram represents the actual number of valence electrons in the form of a dot. Furthermore, it also shows that whether these valence electrons occur in the pair or in the lone form. The chart comes highly recommended to explore and understand the valence electrons in the utmost depth. We highly recommend all chemistry scholars get the dot diagram in the systematic study of this chemical element.
Valency of Lawrencium
Valency is just the other key component for the chemical properties of the element. We also refer to it as the combining capacity of the element which depicts the useful information on the other chemical properties of such element. So, in the case of Lawrencium, we have its valency as the 3 (Three) just like its valence electrons. However, the valence electrons and valency both are different aspects of the element.